Structural analysis of immune cell receptors for an adjuvant that improves the effectiveness of vaccines
Research Press Release | October 25, 2013
| Press Release | ||
|---|---|---|
| Key Points | – The three-dimensional structures of the C-type lectin receptors Mincle and MCL, which bind to glycolipid components as vaccine adjuvants, have been elucidated by X-ray crystallography.
– Mincle and MCL possess hydrophobic regions that are not seen in other similar receptors, which suggest that these regions may be important for binding to glycolipids. Experiments with mutant Mincle confirms the importance of these hydrophobic regions. – The information obtained in this study on the three-dimensional structure of Mincle and MCL have the potential to contribute to the logical design of highly active adjuvants to improve the effectiveness of vaccines. |
|
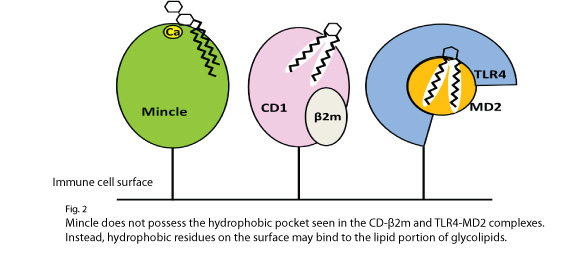
| Overview | Macrophage inducible C-type lectin (Mincle) and macrophage C-type lectin (MCL) are proteins that are expressed on the surface of immune cells. Mincle and MCL have been shown to bind to Mycobacterium-derived glycolipids contained in the well-known BCG vaccine against tuberculosis which activate the immune system and contribute to the removal of pathogens. Mincle and MCL are classified as calcium-dependent (C-type) lectin receptors, but unlike other C-type lectin receptors, they do not bind to adjuvant glypolipids, and the molecular mechanism of this binding was unclear.
The research team used X-ray crystallography to analyze Mince and MCL and determine their three-dimensional structure, with the aim of elucidating this mechanism. Single crystals of Mincle and MCL were produced, and were analyzed by X-ray crystallography. Superimposition of the structures of Mincle and MCL with those of other C-type lectin receptors showed that both Mincle and MCL possessed specific hydrophobic regions that are believed to recognize lipids in the vicinity of the sugar-recognizing site. To ascertain whether or not these hydrophobic regions are actually involved in lipid recognition, mutant receptors were produced with these regions replaced by those of other C-type lectin receptors. The results of binding between these mutant versions and glycolipids showed that their binding capacity greatly decreased. This strongly suggests that what we concluded about the glycolipid-recognizing structure of Mincle was correct. Adjuvants are important for increasing the antibody production capacity of vaccines that are currently under development.The structural information about Mincle and MCL obtained from this study may potentially enable the design of substances with the high immunoenhancing capacity that is essential for vaccines. This study has been published in Proceedings of the National Academy of Sciences of the USA, and was supported by Grants-in-Aid for Scientific Research, JST Strategic Basic Research Programs, and other funding. |
|
| Inquiries |
Katsumi Maenaka, Professor, Faculty of Pharmaceutical Sciences, Hokkaido University TEL:+81-11-706-3970 FAX:+81-11-706-4986 e-mail: maenaka@pharm.hokudai.ac.jp Laboratory of Biomolecular Science (in Japanese) |
|
|
Japanese Link |
http://www.hokudai.ac.jp/news/131008_pr_pharm.pdf | |
| Publications | Proceedings of the National Academy of Sciences U.S.A. (2013.10.7) | |