A Novel Differentiation/Induction Mechanism of Human Immunosuppressor Cells
Research Press Release | October 05, 2015
| Press Release | ||
|---|---|---|
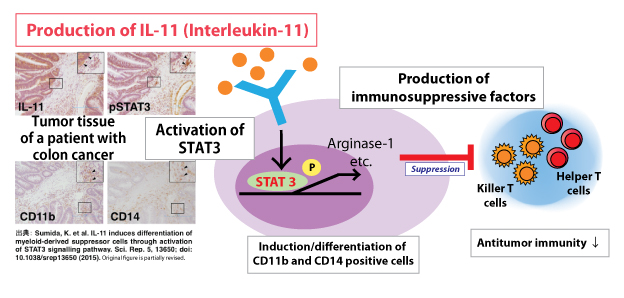
| Key Points | ・We succeeded in the differentiation/induction of human immunosuppressor cells using IL-11.1
・We identified the control mechanism of the immunosuppressive function mediated by STAT3 activation.2 ・Development of new immunotherapies for cancer, which can cancel the immunosuppressive condition of cancer patients, is expected. |
|
| Overview |
Although the life prognosis for cancer patients has significantly improved due to recent progress in the medical field, cancer is still the leading cause of death in Japan. Great effort has been made in the research and development of cancer immunotherapies and the standard of treatment has improved in recent years, but there are still many patients who cannot be helped. This study is the first to discover how IL-11 is produced in the tumor microenvironment of patients with colon cancer, and how cancer cells that suppress the immunoresponse through STAT3 activation are differentiated and induced by peripheral blood lymphocytes. Consequently, by disrupting the signal transmission route of IL-11-STAT3, cancellation and improvement of the immunosuppressive condition of cancer patients is considered possible. Application of this discovery is expected to lead to the development of new cancer immunotherapies. Terms: 1 IL-11: Interleukin-11. A functional protein/cytokine which acts on immune cells such as lymph nodes, monocytes, and macrophages, as well as hematopoietic stem cells, fat cells, and osteoclasts, by stimulating individual cells such as fibroblasts, endothelial cells, and osteoblasts. IL-11 synergistically acts on hematopoietic stem cells along with IL-3 and IL-6. In addition, IL-11 is a multi-functional cytokine which has been previously reported to act on the neural system, inhibit bone formation, and attenuate smooth muscle and heart muscle proliferation. 2 STAT3: A signal transducer and activator of transcription. When cytokines, such as IL-6 and IL-11, and growth factors such as EGF (epidermal growth factor) come in contact with receptors and cells are excited, phosphorylated STAT3 migrates into the nucleus of cells, and promotes the induction of various target genes. STAT3 has been discovered in both immune cells as well as cancer cells, and cell death is induced when its function is suppressed. Due to these characteristics, much attention has been given to STAT3 inhibitors as an anticancer drug which target cancer cells. |
|
| Inquiries |
Hidemitsu KITAMURA, Assoc. Professor, Institute for Genetic Medicine, Hokkaido University TEL: +81-11-706-5520 FAX: +81-11-706-5519 E-mail: kitamura@igm.hokudai.ac.jp |
|
|
Japanese Link |
ヒト免疫抑制細胞の新しい分化・誘導メカニズムが明らかに (2015.9.16) | |
| Publications | IL-11 induces differentiation of myeloid-derived suppressor cells through activation of STAT3 signalling pathway, Scientific Reports (2015.9.1) | |
