A protein that makes skin cancer cells more invasive
Research Press Release | November 21, 2018
Loss of a protein called TRIM29 promotes cancer cell invasion in a common type of skin cancer, suggesting a novel diagnostic marker and a possible therapeutic target.
Squamous cell carcinoma is a common type of skin cancer, usually affecting skin that is exposed to the damaging ultraviolet rays of the sun. A protein that normally helps repair DNA, called TRIM29, is also involved in cancers of the breast, pancreas, prostate, lungs, bladder and stomach. Its role in squamous cell carcinoma, however, was unknown until now.
A research team led by dermatologist Teruki Yanagi and Professor Shigetsugu Hatakeyama at Hokkaido University found that human squamous carcinoma cells with lower levels of TRIM29 were more mobile and invasive, and thus correlated with a worse prognosis. This study was published in Cancer Research.
TRIM29 expression was highest in normal skin cells. It was lower in squamous cell carcinomas, and even lower in tumours that metastasized, migrating from the main cancer site to other parts of the body, such as the lungs. The researchers also determined that turning off the gene made the cancer cells more mobile and invasive, both in cell cultures and mice.
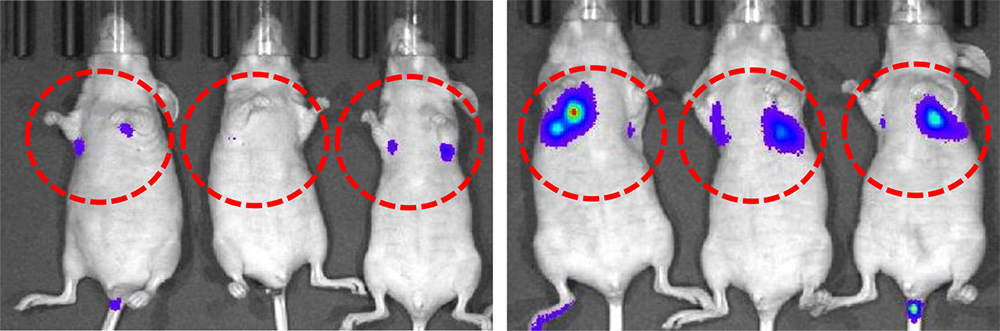
TRIM29 knockdown increases the metastatic potential of human squamous cell carcinoma (SCC). SCC cells were injected to mice with (right) or without (left) knocking down TRIM29 gene. Images were taken 23 days after the injection. (Yanagi T. et al., Cancer Research, November 2, 2018)
Interestingly, they found that TRIM29 interacts with another protein inside cells called keratin, which plays numerous roles in forming a protective structural framework within cells and also in cell motility and signalling.
When TRIM29 levels are normal in a cell, it makes sure keratin is widely distributed within the cytoplasm. On the other hand, when TRIM29 levels are low, keratin distributes mainly around the cell nucleus, resulting in altered cell shapes.
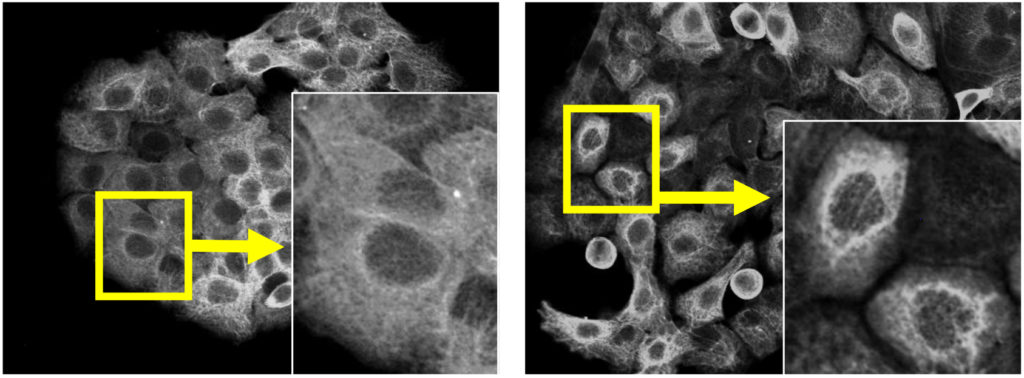
TRIM29 knockdown (right) led to the perinuclear distribution of keratin in human squamous cell carcinoma cells. The left panel shows the control experiment. White: Keratin. (Yanagi T. et al., Cancer Research, November 2, 2018)
Previous studies had shown that TRIM29 suppressed breast and prostate cancer development, because the protein is spread throughout the cytoplasm in these types of cells. But it promoted development of pancreatic, lung, bladder and stomach cancers, where it is localized in the nucleus. In skin cells, TRIM29 is normally positioned throughout the cytoplasm, reinforcing its protective role in suppressing skin cancer.
“Our findings suggest that TRIM29 could be a novel diagnostic and prognostic marker in squamous cell cancers common in skin, neck and head. The TRIM-29/keratin interaction could also be a therapeutic target for treating advanced and metastatic squamous cell cancers,” says Teruki Yanagi.

Dr. Teruki Yanagi (lower right), Dr. Hiroo Hata (lower left), and Dr. Shinya Kitamura (upper left) of the research team at Hokkaido University.
Original article:
Yanagi T. et al., Loss of TRIM29 alters keratin distribution to promote cell invasion in squamous cell carcinoma. Cancer Research, November 2, 2018.
10.1158/0008-5472.CAN-18-1495
Funding information:
This study was supported by KAKENHI (#15H05998, #16K1970106, #18K08259, #17K19506, #18H02607) from the Ministry of Education, Culture, Sports, Science and Technology in Japan, and by the Ichiro Kanehara Foundation for the Promotion of Medical Science, the Cosmetology Research Foundation, the Ono Cancer Research Foundation, the Takeda Science Foundation, the Suhara Memorial Foundation, the Geriatric Dermatology Foundation, the Uehara Memorial Foundation, the Pias Skin Research Foundation, the Life Science Foundation of Japan, the Princess Takamatsu Cancer Research Fund, the Japan Foundation for Applied Enzymology, the Tokyo Biochemical Research Foundation, and the Project Mirai Research Grants.
Contacts:
Specially Appointed Assistant Professor Teruki Yanagi
Faculty of Medicine and Graduate School of Medicine
Hokkaido University
Email: yanagi[at]med.hokudai.ac.jp
Professor Shigetsugu Hatakeyama
Faculty of Medicine and Graduate School of Medicine
Hokkaido University
hatas[at]med.hokudai.ac.jp
Naoki Namba (Media Officer)
Institute for International Collaboration
Hokkaido University
Tel: +81-11-706-2185
Email: en-press[at]general.hokudai.ac.jp

