Using Germline Genome Editing to Prevent the Inheritance of Genetic Diseases: Presentation of Issues for Social Discussion
Research Press Release | June 26, 2015
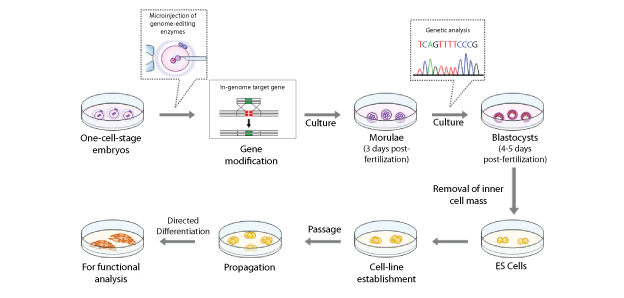
In several countries, for reasons such as improving the techniques for assisted reproduction treatment, human embryos are being produced and culturing them is permitted as far as day 14 or until the formation of the primitive streak. If embryonic stem (ES) cells are established at the blastocyst stage then it is possible to continue culturing and to analyze function. In that case, in Japan it is necessary to comply with guidelines related to human ES cell establishment.
| Press Release | ||
|---|---|---|
| Key Points | ・It is estimated that the feasibility of gene modification in human embryos through genome editing will reach the level of clinical application in the near future.
・In Japan, it is possible to perform human embryo research under regulations, but without sufficient explanation, the research would give cause for societal concern. ・Issues related to genome editing medicine to prevent the inheritance of severe genetic diseases in offspring are presented. |
|
| Overview | Recently, genome editing technology that has spread worldwide has enabled highly efficient modification of targeted genes. However, since the publication of a paper on genome editing of human embryos in April of this year, a worldwide concern has arisen with regard to health damage and the possibility of improper use for non-medical purposes that could affect future generations. Prior to discussing whether to go forward with medicine that involves germline gene modification, it is important to present the bioethical issues, and take into account research trends, existing regulations, and relevant cases.
Last year, we looked at the possibilities for human germline genome editing research and found problems with the related regulations. Now, after a detailed analysis of genome editing research in mammals, we see that gene modification of embryos, spermatogonial stem cells, and oocytes is being conducted using not just mice but also medium- to large-sized animals, and even though there are technical issues such as the introduction of modifications other than at the target sites, we estimate that the level of clinical application will be attained in the near future. We checked into the regulations on research accompanying the creation of human embryos, and found that such research is permitted under regulations in several countries including Japan. We also summarized issues regarding the appropriateness of germline gene editing research, considering topics such as preimplantation diagnosis as well as mitochondrial replacement through nuclear transfer between oocytes or embryos (regulations proposed in England regarding mitochondrial replacement were approved this year). As a result, we found that gene editing research aimed specifically at preventing the inheritance of severe genetic diseases in children could be appropriate. However, an understanding of the ethical status of embryos, and protection of the dignity of the unborn child, will be important issues for the future. For the present, as further improvements are expected in safe genome editing, we propose starting a wider discussion involving the general population. |
|
| Inquiries |
Tetsuya ISHII, Professor, Office of Health and Safety, Hokkaido University TEL: +81-11-706-2126 FAX: +81-11-706-2295 E-mail: tishii@general .hokudai.ac.jp |
|
|
Japanese Link |
生殖細胞系ゲノム編集による遺伝子疾患の遺伝予防:社会的議論のための論点提示 | |
| Publications | Germline genome editing research and its socioethical implications, Trends in Molecular Medicine (2015.6.13) | |