How a cell doubles its DNA matters more than we thought
A new study shows that how a cell fails to divide after copying its DNA can determine its fate.
Cell division is one of the most fundamental and complex processes underpinning life. In human cells, thousands of molecules coordinate with one another in highly precise steps, all within a fraction of a second. But things don’t always go as planned.
Before a cell divides into two, it must first copy its DNA, so that each new cell receives a complete set. Occasionally, what can happen is, a cell successfully copies its DNA but then fails to split into two. When this happens, the cell is left with two copies of its DNA—a condition known as whole genome duplication (WGD).
One way to picture this is to imagine photocopying a document. Normally, you would make two copies and place one in each folder. In whole genome duplication, the copies are made but not separated, leaving one folder with both copies.
Whole genome duplication is not just a minor error; it can influence whether a cell continues to function, becomes inactive or dies, changes its role and differentiates, accumulates errors associated with aging, or contributes to diseases such as cancer.
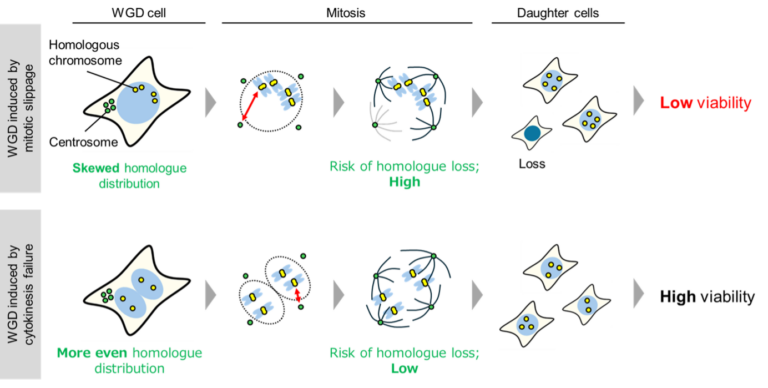
In a new study, researchers at Hokkaido University examined two major ways in which cells fail to divide and undergo whole genome duplication: cytokinesis failure and mitotic slippage.
In cytokinesis failure, the cell completes most of the division process but does not physically split into two due to an error in the cytoplasm. In mitotic slippage, the cell begins dividing but exits the process prematurely without properly separating its genome.
“While whole genome duplication occurs through multiple cellular processes, it has been unclear whether differences in the route affect the characteristics of the resulting cells,” says Associate Professor Ryota Uehara, corresponding author of the study.
Although both processes lead to whole genome duplication, the researchers found that they have very different effects on subsequent cell divisions and viability.
Using live-cell imaging and chromosome-specific labeling, the team compared what happens to cells after genome duplication through these two mechanisms. Cells formed through cytokinesis failure were found to be more stable and showed higher survival rate. In contrast, cells formed through mitotic slippage exhibited uneven chromosome distribution and reduced viability.
The researchers traced this difference to how chromosomes are arranged during the process. In mitotic slippage, chromosomes are often distributed unevenly, which compromises cell survival. In cytokinesis failure, however, chromosomes are distributed more evenly, helping maintain genomic balance and supporting cell viability. When the team experimentally improved chromosome separation in cells undergoing mitotic slippage, their viability was significantly restored.
These findings are particularly important for cancer research. Whole genome duplication is commonly observed in cancer cells, and some anticancer treatments can also trigger it unintentionally. Cells that survive after duplication may continue to grow and contribute to tumor recurrence. The study suggests that targeting processes such as chromosome separation could help limit the survival of these abnormal cells.
“There are different mechanisms through which whole genome duplication can occur, but their distinct impacts have largely been overlooked,” says Uehara. “We challenged this conventional view by comparing cells formed through different mechanisms and found that these differences can influence cell behavior over the long term.”
Original article:
Masaya Inoko et al., Sister chromatid separation determines the proliferative properties upon whole-genome duplication via homologous chromosome arrangement. Proceedings of the National Academy of Sciences (PNAS). April 15, 2026.
DOI: 10.1073/pnas.2524135123
Funding:
This study was supported by JSPS KAKENHI, the Princess Takamatsu Cancer Research Fund, the Kato Memorial Bioscience Foundation, the Orange Foundation, the Smoking Research Foundation, Daiichi Sankyo Foundation of Life Science, the Akiyama Life Science Foundation, the Hoansha Foundation, Sumitomo Electric Group CSR Foundation, and the Terumo Life Science Foundation.
Contacts:
Associate Professor Ryota Uehara
Faculty of Advanced Life Science
Hokkaido University
Email: ruehara[at]sci.hokudai.ac.jp
Tel: +81-11-706-9238
Megha Kalra
Public Relations & Communications Division
Office of Public Relations and Social Collaboration
Hokkaido University
Email: en-press[at]general.hokudai.ac.jp
