Molecular pathway by which stress affects lupus discovered
Research Press Release | July 11, 2022
A novel molecular pathway involving cell signalling proteins in the brain may explain how stress affects neuropsychiatric lupus with diffuse neuropsychological manifestations.
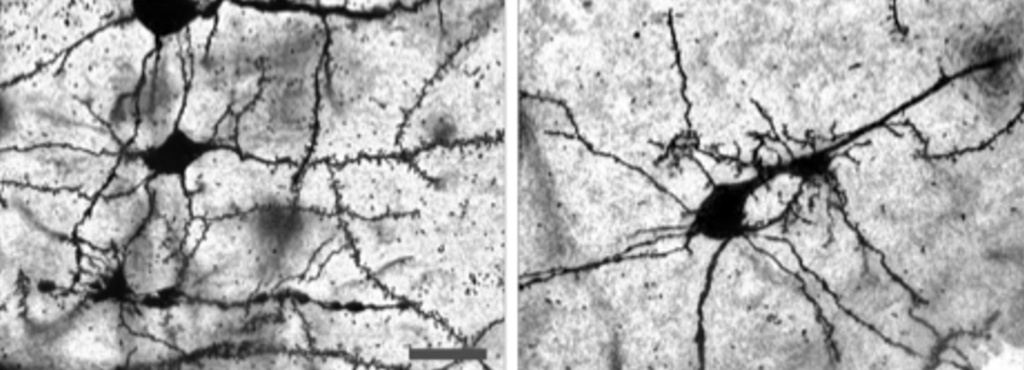
When mice that exhibit SLE-like symptoms are subjected to sleep deprivation stress, their neurons exhibit abnormal growth (left). When IL12 and 23 are blocked, the abnormal growth is reversed (right; Nobuya Abe, et al. Annals of the Rheumatic Diseases. July 11, 2022).
Systemic lupus erythematosus (SLE) is an autoimmune disease that affects all the organ systems of the body. This chronic disease includes periods of illness, called flare ups, and periods of remission, when symptoms are few. There is so far no cure for this disease; treatments involve preventing flare ups and reducing their severity and duration. One of the most severe forms of SLE is neuropsychiatric systemic lupus erythromatosus (NPSLE), which affects the central nervous system—the brain and spinal cord.
A team of researchers, led by Professor Masaaki Murakami at the Institute for Genetic Medicine, Hokkaido University, have identified a stress-induced molecular mechanism that affects NPSLE, revealing a potential target for the treatment of the disease. Their findings were published in the journal Annals of the Rheumatic Diseases.
The research team focused on a specific type of NPSLE called Neuropsychiatric lupus with diffuse neuropsychological manifestations (dNPSLE). There are believed to be many causes for dNPSLE, but its pathogenesis remains poorly understood. The researchers were most interested in the effects of stress, as chronic stress is known to be involved in the development of many autoimmune diseases.
In order to identify stress-induced molecular mechanisms that might affect dNPSLE, they conducted experiments on mice models that exhibit SLE-like symptoms. After subjecting a set of these mice to sleep deprivation stress, they were able to identify that the medial prefrontal cortex (mPFC) of the brain was abnormally activated.
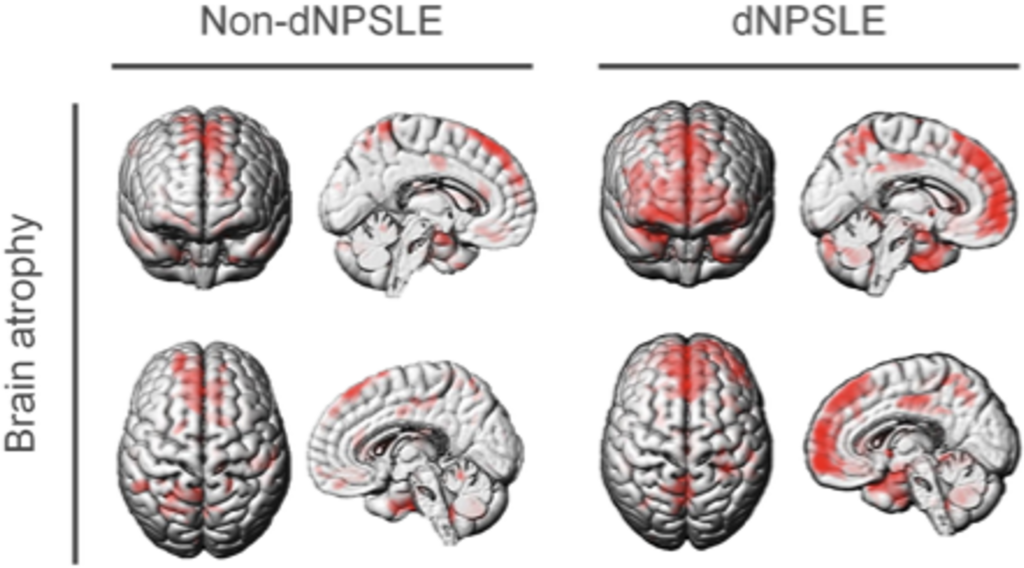
Brains of patients with dNPSLE show atrophy of the medial prefrontal cortex (mPFC, in red) compared to the reference brain of healthy individuals (Nobuya Abe, et al. Annals of the Rheumatic Diseases. July 11, 2022).
In the mPFC, the expression of at least 509 genes was significantly affected by sleep deprivation. The team specifically noted the upregulation of a microglial proinflammatory gene that is required for two interleukins, IL12 and IL23. Further, they showed that upregulation of these two interleukins caused activation of the microglial cells of the mPFC. Blocking IL12 and IL23 pathways in these sleep-deprived mice models inhibited the stress-induced neuropsychiatric symptoms.
Most importantly, they showed that levels of IL12 and IL23 in the cerebrospinal fluid of human patients with dNPSLE were higher than that of healthy individuals, to such an extent that it can be used as a diagnostic marker. They also showed that the mPFC in dNPSLE patients is atrophied. Together, these indicate that the findings from the mice models may be applicable to humans.
“In revealing the effect of the stress-induced effects on the expression of IL12 and IL23 in dNPSLE, we have identified them as not only a diagnostic marker but also a novel therapeutic target for this disease,” said Masaaki Murakami.

The Murakami group at Hokkaido University. Masato Tarumi (last row, third from left; navy blue scrubs) and Masaaki Murakami (first row, center; black zip-up hoodie) are key contributors to the paper (Photo: Masaaki Murakami).
Original Article:
Nobuya Abe, et al. Pathogenic neuropsychiatric effect of stress-induced microglial interleukin-12/23 axis in systemic lupus erythematosus. Annals of the Rheumatic Diseases. July 11, 2022.
DOI: 10.1136/ard-2022-222566
Funding:
This work was funded by grants from the Bristol Myers Squibb Foundation; the Japan Society for the Promotion of Science (JSPS) KAKENHI; the Ministry of Education, Culture, Sports, Science and Technology (MEXT) Q-Leap (JPMXS0120330644); the Japan Agency for Medical Research and Development (AMED); the Joint Usage/Research Center, Institute for Genetic Medicine, Hokkaido University; the Photo-excitonix Project at Hokkaido University; and the Promotion Project for Young Investigators at Hokkaido University.
Contacts:
Professor Masaaki Murakami
Division of Molecular Psychoneuroimmunology
Institute for Genetic Medicine
Graduate School of Medicine
Hokkaido University
Tel: +81-11-706-5121
Email: murakami[at]igm.hokudai.ac.jp
Sohail Keegan Pinto (International Public Relations Specialist)
Public Relations Division
Hokkaido University
Tel: +81-11-706-2185
Email: en-press[at]general.hokudai.ac.jp
Related Press Releases:
Chronic stress induces fatal organ dysfunctions via a new neural circuit
HU Research on Gravity and Inflammation Response Selected for JAXA Feasibility Study
Neural Signal Due to Pain Induction Develops Relapse of a Multiple Sclerosis Model!

